Nihon Kohden Launches Life Scope E7 in India Following Regulatory Clearance
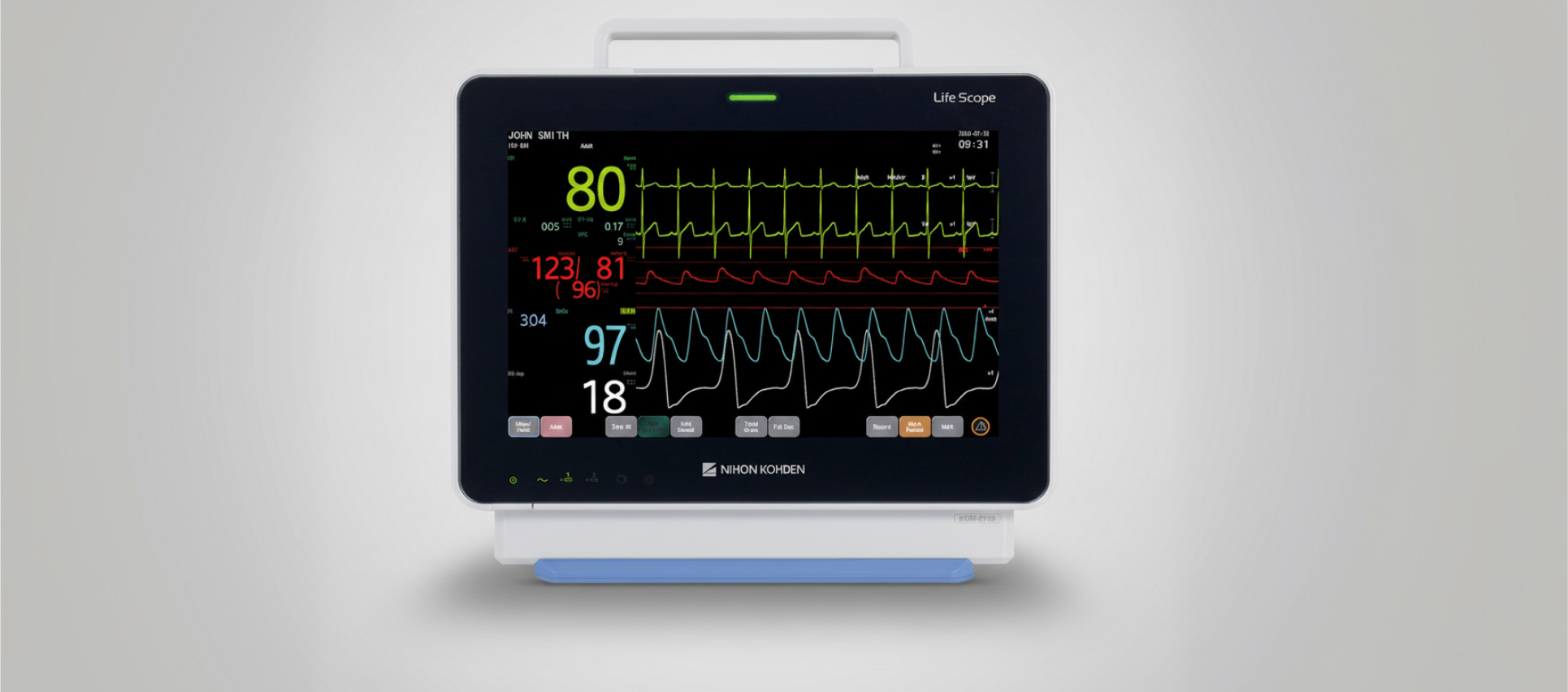
The Life Scope E7 patient monitor consolidates multiple monitoring functions, including ECG, oxygen saturation, invasive and non-invasive blood pressure, temperature, CO2, anaesthetic gases, and neurological parameters such as EEG and aEEG.
Nihon Kohden, a Japan-based medical electronics manufacturer, has introduced the Life Scope E7 in India, expanding access to an advanced multi-parameter monitoring system designed for use across intensive care, emergency departments, operating rooms, and general wards.
The launch follows regulatory approval earlier this year and is scheduled officially for April 16, 2026. And, the rollout comes after the Central Drugs Standard Control Organisation granted import license approval in February 2026, enabling formal market entry.
Yuya Kawabata, Managing Director, Nihon Kohden India, said: "The Life Scope E7 has been developed with a clear design intent — compact form, ease of use, and uncompromising reliability for clinicians working under pressure. We are proud to bring this globally proven platform to Indian hospitals and to contribute to better patient outcomes in critical care."
Globally, the system is already operational across hospitals in Europe, the Americas, the Middle East, Southeast Asia, and Africa, making this its first expansion into South Asia.
The Life Scope E7 patient monitor is available in 12.1-inch and 15.6-inch touchscreen configurations, offering flexibility across care settings. It consolidates multiple monitoring functions, including ECG, oxygen saturation, invasive and non-invasive blood pressure, temperature, CO2, anaesthetic gases, and neurological parameters such as EEG and aEEG.
By combining these capabilities into a single system, the device reduces reliance on multiple standalone machines within hospitals.
The platform also emphasizes data continuity and clinical decision support. It can store up to 120 hours of patient trends, display multiple waveforms simultaneously, and maintain extensive alarm histories, allowing clinicians to retrospectively review patient conditions.
Its proprietary technologies, including cap-ONE CO2 sensing, inflation-based blood pressure measurement (iNIBP), and Pulse Wave Transit Time-triggered monitoring, are designed to improve speed and accuracy in critical care environments.
SynECi18 further enhances cardiac monitoring by generating additional diagnostic views from a standard ECG setup.
Furthermore, the monitor supports integration with ventilators and anaesthesia machines while enabling connectivity with hospital information systems through HL7 protocols. It also uses certificate-based encryption to secure patient data transmitted across networks.
A key aspect of the device is its Human Machine Interface approach, with in-house development of sensors and signal processing aimed at maintaining consistent accuracy and usability.
This design philosophy is intended to support clinicians working in high-pressure environments where reliability and speed are essential.
Stay tuned for more such updates on Digital Health News
































